What are Nanomaterials?

Presented by


Nanomaterials: 1-100 nm
- Nanomaterials have at least 1 dimension measuring less than 100 nm.
- They are generally man-made, inorganic materials.
- To explain, lets look at carbon-based nanomaterials.



Buckyballs
Buckyballs are spherical nanomaterials with all three dimensions confined at the nanoscale.
- Measuring 1 nm in diameter.
- Carbon atoms in buckyballs have the same structure of pentagon and hexagon faces as a soccer ball.
- They are probably the only nanomaterial with a defined molar mass (of 720 g/mol).



Buckyballs
- Buckyballs were the first. carbon-based nanomaterial
- Discovered in 1985 by James R. Heath, a grad student at Rice University.
- 11 years after, in 1996, the professors involved shared the Nobel Prize in Chemistry for this discovery.

Sample application:
- used in next-generation organic solar cells.
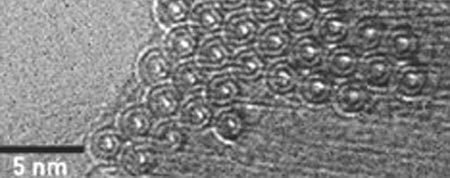
Microscopy of buckyballs
Carbon Nanotubes
- Cross section or diameter is ~1-3 nm.
- They are a pipe 1 atom thick.
- However, nanotube length can be quite long exceeding a micrometer.
Characteristics:
- 150 times stronger than steel
(1/6 of the weight). - Flexible.
- Good conductors of electricity and heat.
- Diameter: 1-3 nm.


Carbon nanotubes are tubular / cylindrical nanomaterials with
2 dimensions at the nanoscale.
Carbon Nanotubes
- Discovered by Sumio Iijima of Nippon Electric Company (NEC), Japan, in 1991.
- It was an accident!: They were trying to make buckyballs but instead observed unusual carbon fibers which turned out to be nanotubes.
- Under microscope they look like hair, thin fibers, or a bowl of spaghetti.


Sample application:
- used in application of flexible electronic displays, next generation of transistors, solar panels, stealth paint (cloaking devices), cancer treatment/therapy.
Graphene
- It is a sheet only 1 carbon atom thick.
- Lateral dimensions can be in the micro or even in the millimeter scale.
Characteristics:
- Excellent conductors of electricity and heat.
- Extremely strong.
- Flexible.
Graphene is a planar nanomaterial with 1 dimension confined at the nanoscale.


Graphene
- Graphene is a derivation of graphite (pencil lead).
- Graphite is multiple layers of graphene stacked on top of each other.
- “Re-discovered” by A. Geim and K. Novoselov in 2004, two researchers at the University of Manchester.
- Developed a practical “scoth tape” technique to pull apart the layers, isolating individual sheets of graphene from bulk graphite.
- Won the 2010 Nobel Prize in Physics.


Sample application:
- Applications are very similar to those of carbon nanotubes.
- Also will be used for flexible displays, sensor devices, high-performing batteries, etc.

Carbon-based Nanomaterials
Nanotubes, graphene, and buckyballs are new allotropic forms of carbon.
-
What is an allotrope?
- Different structural arrangement of the same element resulting in different forms or materials.
- Allotropes of Carbon:
- Diamond
- Graphite
- Amorphous (coal)
- Buckyballs
- Nanotubes
- Graphene
- Carbyne
All of these are made entirely of carbon atoms, but different structural arrangement of the atoms result in different materials
carbon-based
nanomaterials

Carbon-based Nanomaterials
Structural difference give the new, nano allotropes different properties from other forms of carbon. Example:
-
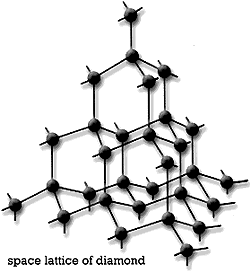
In diamond all carbons are bound to 4 other carbon atoms, but our nano-material structures have carbon bound to 3 other carbon atoms.
-
By only being bound to 3 other C's, the nano-based materials have C atoms with a free electron pair - this is what makes nanotubes and graphene good conductors of electricity vs. diamond.

1
1
2
2
3
3
4
Structure of Diamond
Structure of graphene, carbon nanotubes, and buckyballs
Where Can I Find Them?
- Nanomaterials can be purchased online, like everything else.
- Ex: Carbon Solutions offers a variety of nanotubes and graphene.
-
Compare pricing: 1 gram of purified nanotubes = 7x's more than gold;
1 gram of functionalized nanotubes = 25x's more than gold. -
We need better processes to make the materials cheaper!
- Graphene for example, could be used in tons of applications if only there was a good, cost-effective way to make lots of it.


Carbyne
- Carbyne is the 4th type of carbon-based nanomaterial, but so far is only theoretical.
- Carbyne is a single chain of double-bonded carbon atoms:

- Been studied through simulations & computations.
- It is predicted to be the strongest material ever.
- but, we cannot figure out how to make it.
- We need someone to figure out the mystery!

Model of a carbyne rope
Carbyne is predicted to be shaped as a rope structure
Non-Carbon Nanomaterials
- Carbon-based nanomaterials are unique and often talked about in their own category.
- There are many other types of nanomaterials made from other elements or molecules.



Nanowires
Nanowires = “wire-like” structures with 1-100 nm cross-section.
-
Made of copper, metals, metal oxides.




Examples of copper nanowires:
Sample application:
- Electronics, fundamental components (super-capacitors, transistors), sensor technologies.
Bulk copper wire
Nanoparticles
- Have been made from all kinds of materials.
- Metals, metal oxides (e.g., gold, silver)…
- Have a variety of structures
- Nano-”shells” or nano-”onions” (with different layers of materials).
Nanoparticles are small aggregates/clusters of atoms with dimensions between 1-100 nm.
- They might be 1-2 nm in diameter up to much larger structures (100 nm in diameter or more…).

Nanoparticles
Generally spherical in shape but, but can have many different faces depending on the size.
- The smaller, the more faces it tends to have (more polygonal, less smooth).
- The larger the structure, the smoother it tends to be.
Sample application:
- Depends on nanoparticle composition
- anti-bacterials, personalized medicines, cosmetics, molecular imaging in radiology, and more...

Overview of Nanomaterials: Carbon

- Buckyballs- soccer ball-shaped carbon molecules.
- Nanotubes- pipe- or tube-like carbon structures, 1-atom thick and hollow inside.
- Graphene- a flat, continuous sheet of carbon atoms, 1-atom thick.
- Note: this table does not include carbyne, which is currently only theoretical.
Overview of Nanomaterials: Other
- Nanowires- wire-like materials.
- Nanoparticles- quasi-spherical clusters of a small number of atoms.
- Nanowires and nanoparticles are typically formed from:
- Metals - Iron, Gold, Silver, Nickel, etc.
- Metal oxides - Titanium dioxide, Iron oxide, etc.
- Semiconductors - Silicon, Indium phosphide, etc...

Learn More: Videos to Watch
Carbon Nanotubes: Watch how nanotubes are made and some interesting properties they have as they are played with in the lab.
Learn More: Videos to Watch
Graphene:
The science that makes it so special.
Graphene:
Applications of the next revolutionary material.
In-Class Assignment
- Name the 3 most common types of carbon-based nanomaterials?
- What is a typical diameter of single-walled carbon nanotubes? And buckyballs?
- Nanoparticles can be made of…..
- Referring to the Periodic Table and what you know about nanoparticles, name 3 elements that are not typically used to make nanoparticles.
- How many orders of magnitude does the size of nanoparticles span?
- Nitrogen nanowires are common. True or False?

This lesson was presented by:
To learn more about nanotechnology, visit omninano.org

